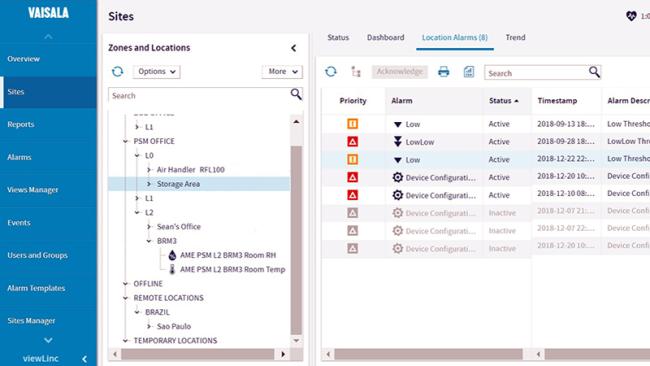
Laboratory Monitoring
Vaisala solutions monitor and measure various laboratory environments including pharmaceutical and biotechnology applications, research and development, equipment calibration, testing and manufacturing.
Vaisala offers the widest array of data loggers, transmitters, probes, and software. For in-line liquid measurements, learn more about Vaisala inline process refractometers.
With nearly unlimited parameters, our solutions provide reliable measurements for almost any lab application and are ideal for Process Analytical Technology (PAT) methods, Good Laboratory Practice (GLP), and Good Clinical Practice (GCP) guidelines.
Vaisala sensors and systems ensure consistency in pharmaceutical and biotechnology components, including processing equipment and enclosures, safeguard the quality of in-process and finished products, maintain accurate records, and provide analytics for review and audits.
For both GMP and other critical laboratory applications, Vaisala sensors and systems provide accurate data, complete and compliant records, and a full suite of services, including calibration, installation, validation, and technical support.
Laboratory Monitoring & Measurement Applications
- Refrigerators, freezers, cold rooms, LN2
- Research & Development
- Cleanrooms
- Calibration laboratories
- Incubators
- Stability testing chambers
- Glove box, isolator, transfer hatch bio-decontamination
- Drying, compressed air
- Animal facilities
Laboratory Environmental Monitoring
Comprehensive monitoring in a secure cloud
Jade smart cloud provides access to real-time measurement data, historical trends, and data visualization with intuitive dashboards. Generate reports effortlessly, monitor environments both locally and remotely, and ensure data integrity with secure, encrypted communication and certificate-based device authentication.
Audit-ready & GxP-compliant monitoring
Stay in control with real-time data, historical trend analysis, and simplified validation. viewLinc offers 21 CFR Part 11/Annex 11 compliant reporting, secure remote and local access, and industry-leading accuracy.
What customers say
“Originally we installed the [monitoring] system in our ultra-low temperature freezers and a cryogenic freezer. After installing the loggers in freezers, we installed more data loggers in the pharmacy. Later we expanded the system to our refrigerators and to monitor ambient temperature and humidity.”
Dr. Leonardo Motta - University of Caxias do Sul
“The simple Web-based operation, access for all users, as well as automated printouts of reports were important advantages for us. The system-supervising lab technicians no longer have to send manual reports, as they are now created and sent automatically, leaving more time for core scientific tasks.”
Ralf Freese, Medical Director, Director, Business & Corporate Development at CTC North
"[Vaisala's] sensors and recorders are easily interchangeable and moveable so that the two parameters can be measured in the chambers or the rooms in which they are installed... our array of [data loggers] is flexible and can be spread out between one or more breeding chambers or one or more experimentation rooms."
Prof. David Siaussat, Institute of Ecology & Environmental Sciences, Sorbonne University
Laboratory Monitoring & Measurement Solutions

viewLinc Monitoring, Alarming and Reporting Software
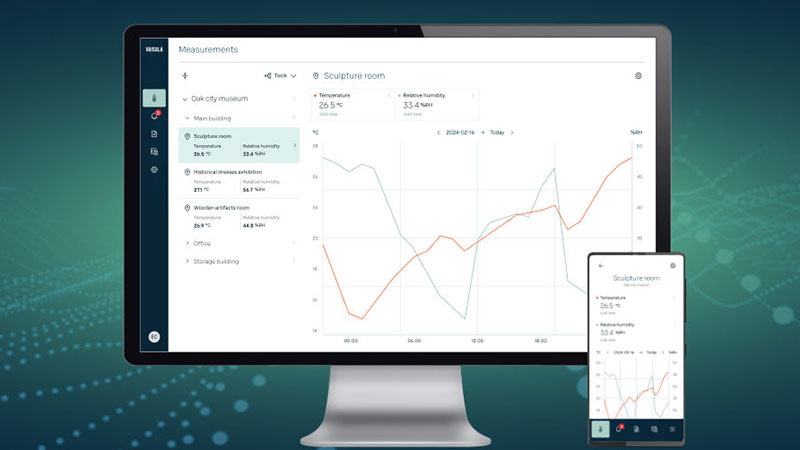
Jade Smart Cloud

VaiNet Wireless Temperature & Humidity Data Logger RFL100

VaiNet Wireless Temperature Data Logger RFL100

VaiNet wireless CO2 data logger RFL100

VDL200 Data Logger
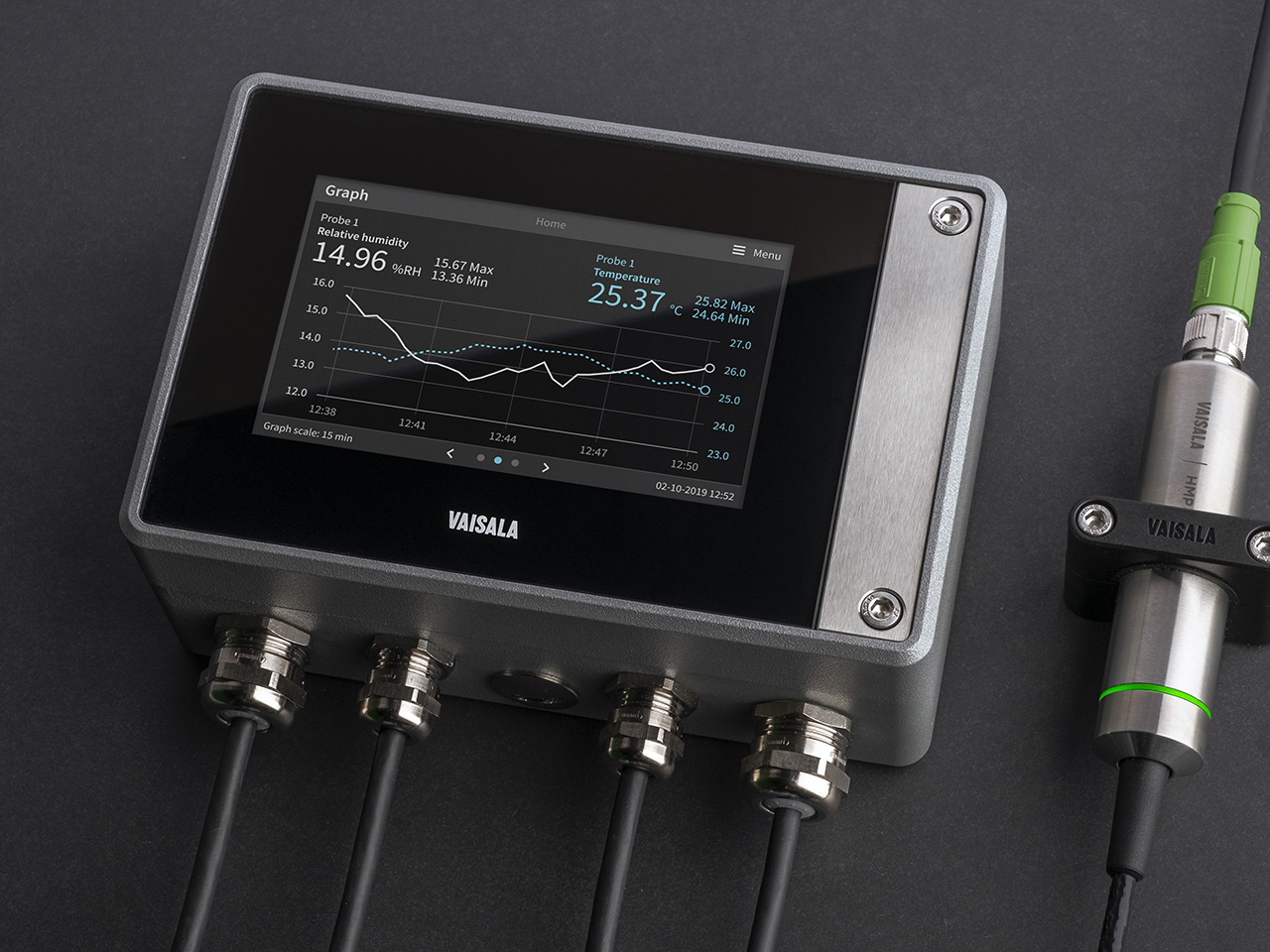
Indigo500 Series Transmitters

Indigo200 Series Transmitters for Vaisala smart probes

Indigo80 handheld measurement device

GMP80P portable carbon dioxide probe with pump sampling

CO₂ Probe GMP251

CO₂ Probe GMP252

Temperature Transmitters TMT120/130