viewLinc Continuous Monitoring Self Study Center
The science behind monitoring and alarming for GxP regulated environments
As a company, Vaisala is always looking to help you the consumer understand the science behind our products. In our Continuous Monitoring Self Study Center, learn the fundamentals of monitoring for GxP regulated environments, GAMP principles in monitoring system validation, insights to data integrity, best practices for controlled environments and wireless monitoring. Vaisala is founded on 85 years of experience, know how that experience can help you in solving your most difficult life science questions.

Monitoring System Validation using GAMP Methodology
Good Automated Manufacturing Practices (GAMP) what are they? Avoid unwanted observations and issues during customer audits by utilizing GAMP methodology as a reasonable and systematic guide to ensure your monitoring system software performs as expected throughout its lifecycle. Review the whitepaper on GAMP and learn to Validate CMS Software using GAMP principles from our Regulatory Expert.
- Webinar: Validating CMS Software: Using GAMP principles in monitoring system validation
- White Paper: Using GAMP Methodology to validate environmental monitoring system software
- Infographic: Take 10 steps with you

21 CFR Part 11 & Annex 11 for Monitoring Systems
You may have to know only one or the other depending on the location of your GxP application. Regulatory changes come in waves, make sure that you are up to date with our two application notes and webinar on 21 CFR Part 11 & Annex 11 for Monitoring Systems.

Data Integrity and Monitoring
Discover the 8 recommendations for establishing and maintaining good practices for data integrity while expanding upon
- Recent guidance and documents
- Evaluating data integrity risks
- Pitfalls of data management inGxP applications
- Best Practices for ensuring and preserving data integrity.
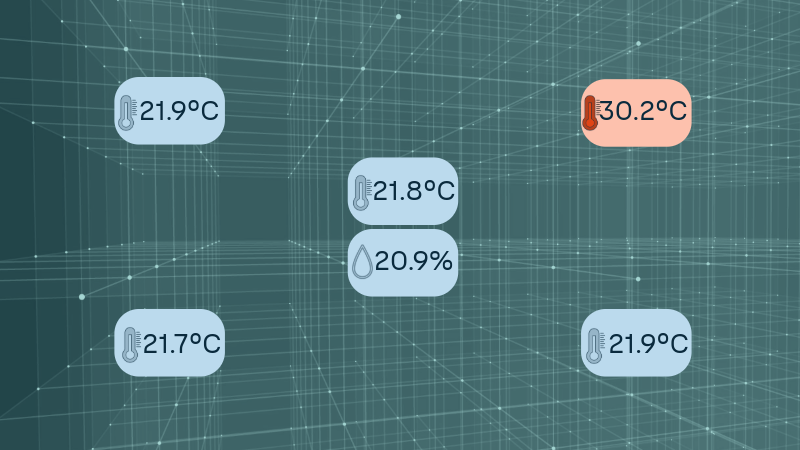
Mapping Best Practices for Controlled Environments
Temperature and humidity qualification for GxP environments is not a difficult task especially when done correctly. Senior Regulatory Compliance Expert Paul Daniel provides you with a validation mapping kit, two webinars, and a Step-by-step guideline for Validating Life Science Storage Facilities to ensure you get the job done right the first time.

Wireless Monitoring with VaiNet Technology
When you combine monitoring for GxP and controlled environments with wireless technology, naturally there are a lot of questions. To list a few:
- What kind of signal ranges are now achievable?
- What wireless features ensure a secure signal?
- What kind of wireless structure is effective?
- What is the infrastructure investment?
- How many data loggers per single network access point?
Get the answers to all of those and your questions by familiarizing your team with our webinar application note and informative videos.
Latest and Greatest
New Covid-19 Vaccine Benefit from tried and true temperature monitoring
Proper storage and distribution of Covid-19 vaccines are key to conquering the virus
CMS Products
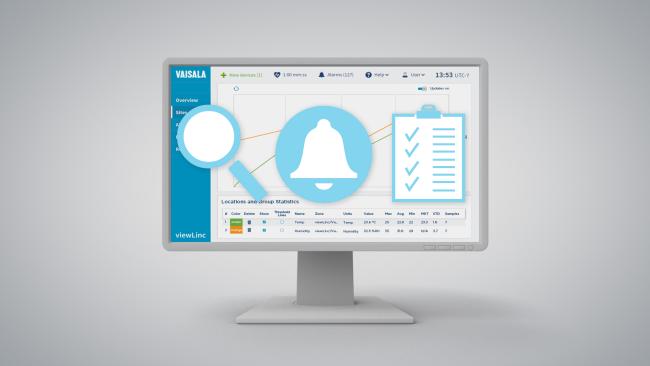
viewLinc
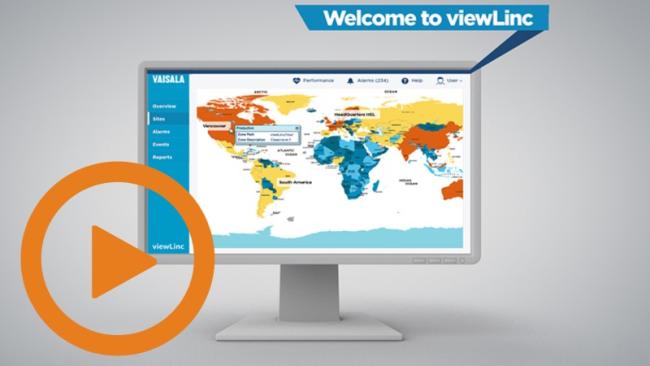
A simple overview

viewLinc News
Dive into the latest and greatest from viewLinc. Explore Vaisala's most recent webinars, whitepapers and product developments that will keep you at the forefront of your next GxP regulated environment conversation.
Questions? contact our Continuous Monitoring Team.
We will respond within 24hrs.


