Temperature, Condensation Point & Maximum Achievable vH2O2 ppm in Bio-decontamination
This blog is the third in a 4-part series where we describe how process parameters influence vaporized hydrogen peroxide bio-decontamination applications.
- First blog: Humidity
- Second blog: Solution Concentration
In this series, we propose four basic process parameter rules. This blog focuses on the third rule:
Increasing the temperature increases the amount of water and hydrogen peroxide vapor the air can hold, thereby increasing the maximum achievable vH2O2.
In our previous blogs in this series, the bio-decontamination cycle temperature was maintained at 23 °C for all exemplar cycles. At a given temperature, air can only hold a certain amount of vapor, whether H2O or H2O2. Now will will demonstrate how changing the temperature can change both the condensation point and the maximum achievable hydrogen peroxide vapor concentration.
Figures 3a and 3b (below) show two bio-decontamination cycles. The cycle represented by black lines indicates a process temperature of 40 °C and the blue lines represent temperature at 23 °C. In both cases, dehumidification is performed to lower humidity to 10% prior to conditioning and both use the same H2O2 solution concentration (59 m-%). The black line in Figure 3b shows that temperature at 40 °C allows a higher ppm vH2O2 than temperature at 23 °C.
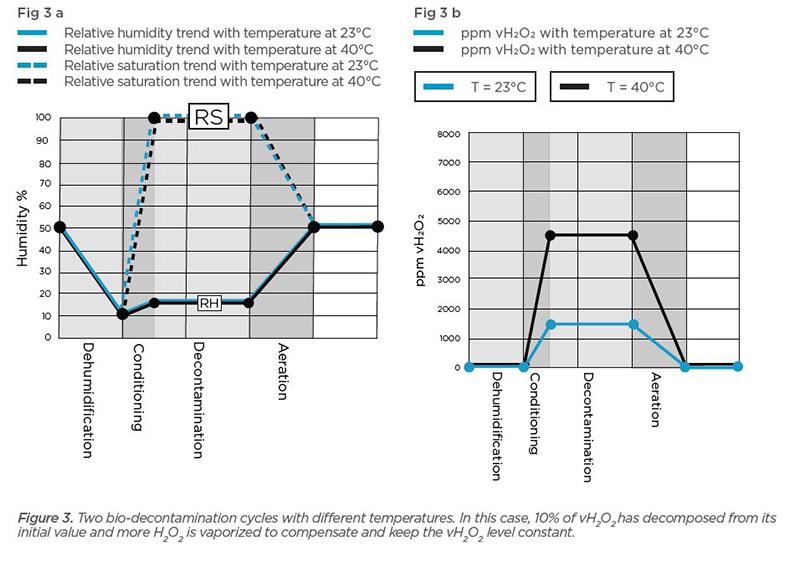
We can further examine the effect of temperature by looking at humidity, both relative humidity and relative saturation values. (Read about relative saturation.)
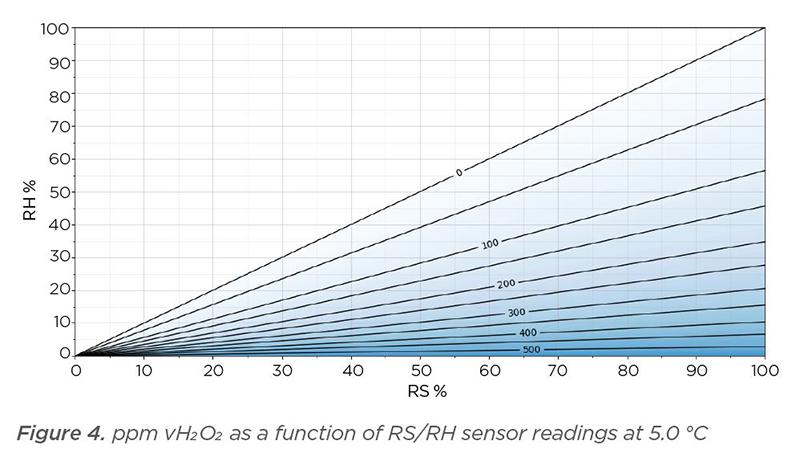
Figure 4 below shows ppm vH2O2 with temperature at 5°C. Relative saturation is on the x axis and relative humidity is on the y axis. The coordinate lines within the x and y axes represent H2O2 vapor concentration from 0 ppm to around 500 ppm. The 0-ppm line represents a vapor that has been created using pure water only. When the H2O2 solution concentration is increased, the vH2O2 also increases. Theoretically, the line along the X-axis represents a vapor that has been created using 100% H2O2 liquid. Condensation occurs once relative saturation is 100% RS and ppm vH2O2 cannot be increased thereafter. Thus, with temperature at 5 °C, initial relative humidity at 0% RH, and vaporizing an H2O2 solution of 100 m-%, the theoretical maximum achievable ppm vH2O2 is 548 ppm.
All conditions in Figure 5 are the same as Figure 4 (0% RH, 100 m-% solution), and only temperature is changed to 50 °C. The theoretical maximum achievable ppm vH2O2 is now 13,019.
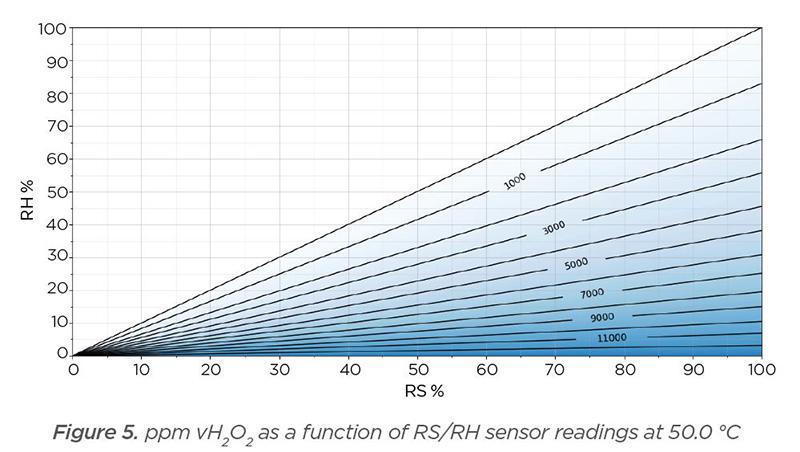
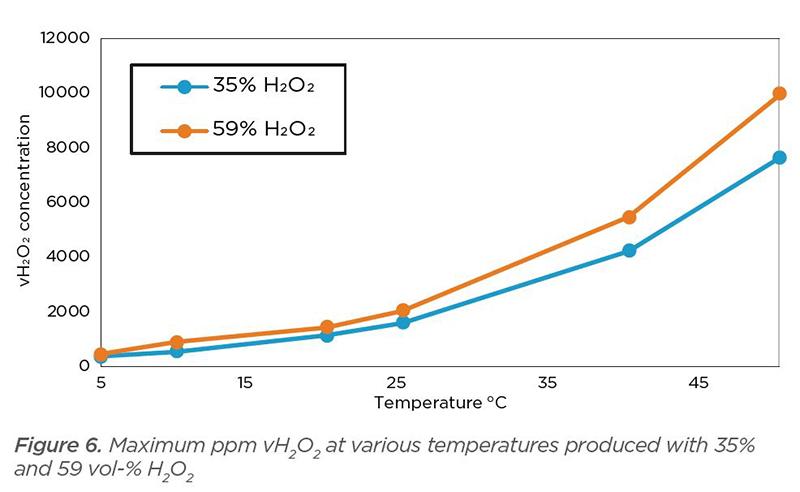
Figure 6 shows the maximum ppm vH2O2 at various temperatures and using different H2O2 solution concentrations. We compare two frequently used H2O2 solution concentrations: 35% and 59%. The blue trend line represents a 35% H2O2 solution. With temperature at 40 °C, the maximum achievable ppm vH2O2 is 4,210. At the same temperature (40 °C), a 59% H2O2 solution gives a maximum achievable ppm vH2O2 of 5,461.
Read about all four rules in the white paper: Considering Condensation: Influences in Hydrogen Peroxide Vapor Bio-decontamination
We invite you to view a recorded webinar on this topic.
Please feel free to add comments and questions in the fields below or contact us.




Add new comment