Create a validation master plan
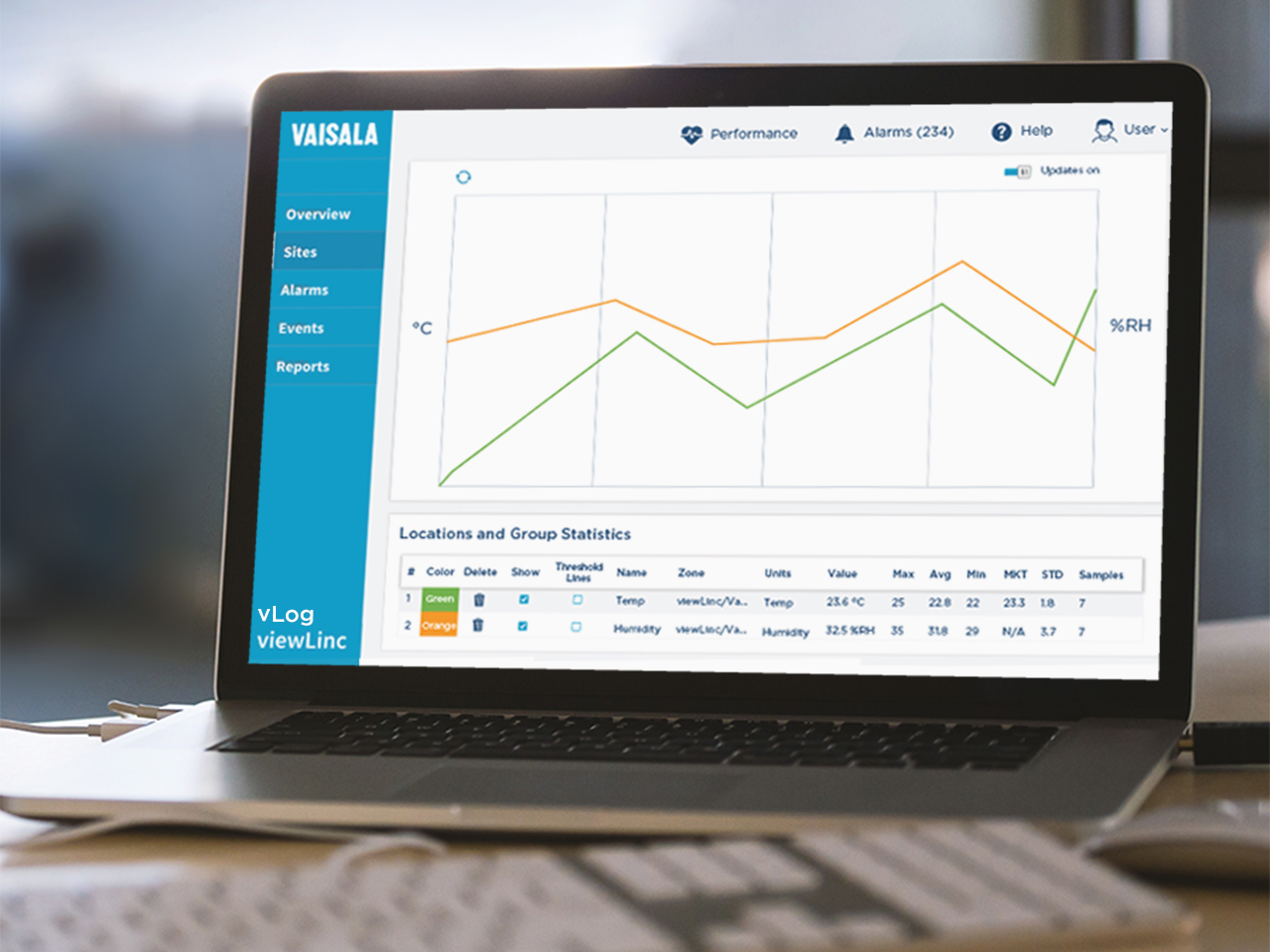
Establish a validation plan that takes a risk-based approach. Where will environmentally sensitive products and materials be stored?

Reduce the risk of non-compliance and lost products in your life science holding and storage applications areas with the viewLinc Continuous Monitoring system:
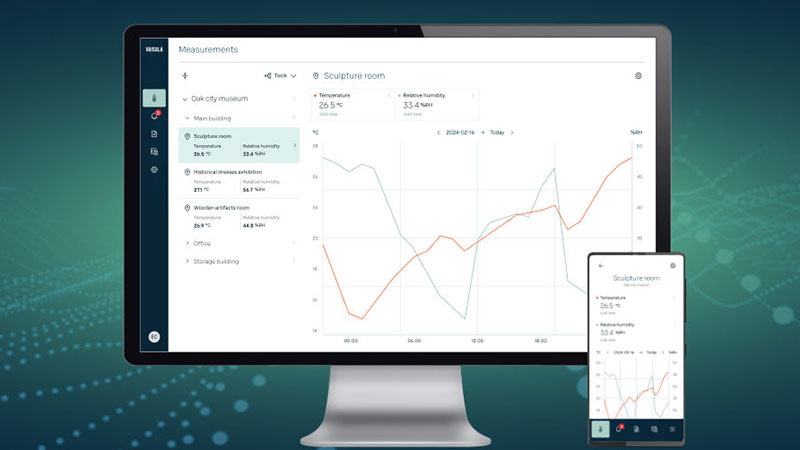
Jade Smart Cloud monitoring system ensures the quality of stored goods, such as electronics, perishable goods, food & beverage, high-quality mechanical and aerospace products and other valuable items:
Establish a validation plan that takes a risk-based approach. Where will environmentally sensitive products and materials be stored?
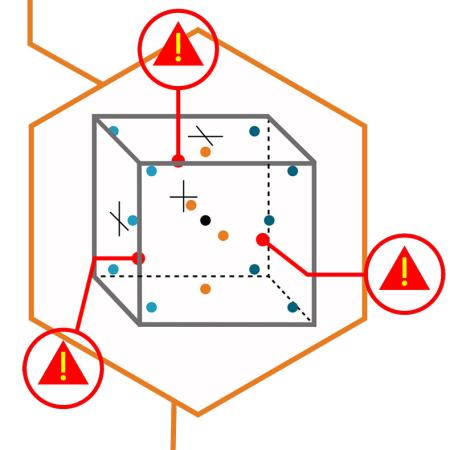
Identify risk from variations in temperature and humidity. Account for :

Based on identified risks, make decisions on what your data needs are for your mapping study.
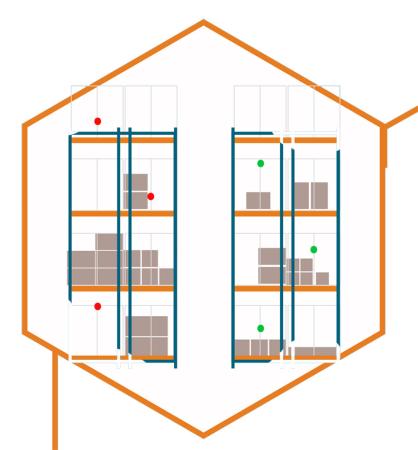
How can we create a uniform distribution of sensors that will give us a full understanding of the environment? Take into consideration external walls, windows, lighting, and any type of air circulation. Storage shelf capacity has an effect on airflow and temperature gradients. Will humidity have an adverse effect on products? Consider using both temperature and humidity sensors.

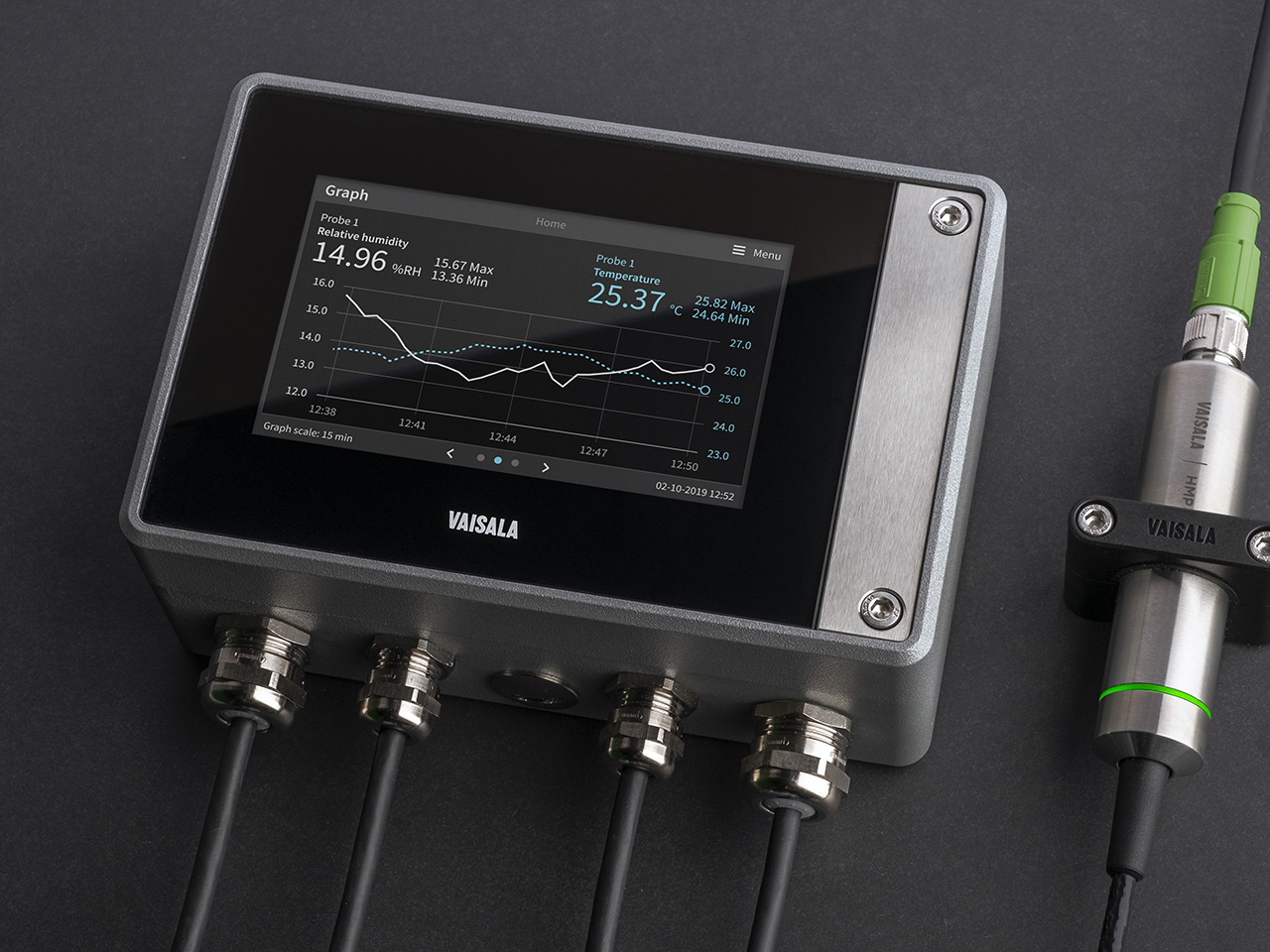
Choose equipment designed for mapping studies.
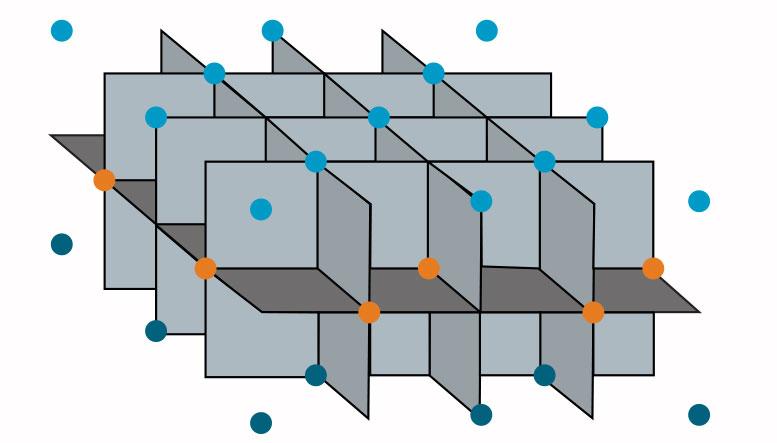
Setup sensors where planned
Conduct a test and look at data for these points:
Identify locations where products may be exposed to unacceptable conditions, then make adjustments to storage locations. Don't forget to modify your validation protocol in light of results from initial mapping test.
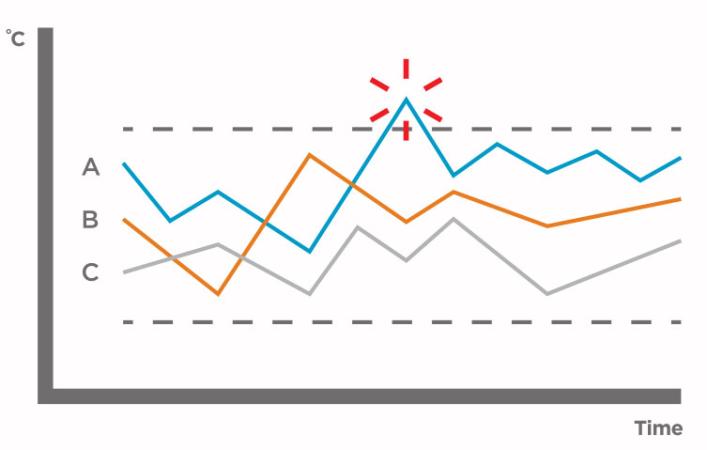
Conduct and document a mapping study for approval then plan out mapping schedule.