
冷蔵倉庫
冷蔵倉庫は、冷蔵環境を必要とする生鮮食品の品質を維持します。冷蔵倉庫の適正な湿度制御は、霜の蓄積を低減し、エネルギー効率を向上させ、解凍コストを削減します。
ヴァイサラの湿度温度変換器ポートフォリオには、相対湿度、露点、霜点などのさまざまな湿度パラメータを計測する製品を、広範な動作温度範囲で取り揃えています。
関連製品

TMP1 温度プローブ
ヴァイサラ TMP1 温度プローブは、精度や堅牢性が不可欠な製薬業界や校正試験所など、温度計測に厳しい要件が求められる産業用途向けに設計されています。
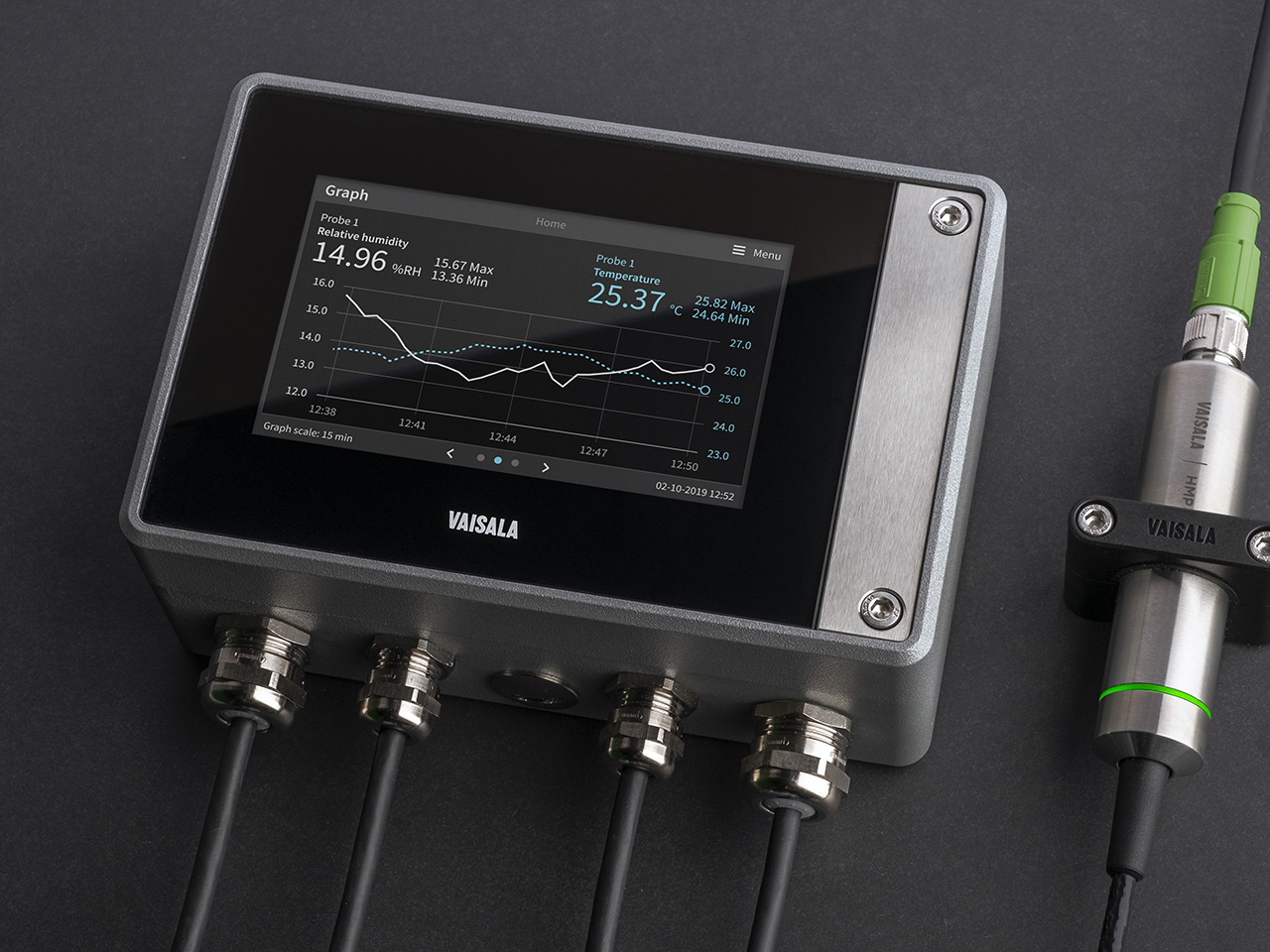
Indigo500シリーズ 変換器
ヴァイサラIndigo500シリーズ 変換器は、ヴァイサラIndigoシリーズのスタンドアロン型スマートプローブ用のホストデバイスです。Indigo500シリーズには、多機能のIndigo520 変換器と、基本機能を備えたIndigo510 変換器があります。

Indigo200シリーズ変換器:ヴァイサラスマートプローブ用
ヴァイサラIndigo200シリーズ 変換器は、ヴァイサラの湿度、温度、露点、オイル内水分、CO 2 、H 2 O 2 用スマートプローブの計測値を表示するホストデバイスです。

viewLinc モニタリング、アラーム通知、レポート作成ソフトウェア
viewLinc Enterprise Server ソフトウェアは、温度、相対湿度、CO2、差圧、高低差、ドア接点など、多数のパラメータのモニタリングが可能で、既存ネットワークへの接続の容易性、リモートアクセス性、モニタリングポイントを数千まで追加できる拡張性によって、機器の所有コストを低減させています。
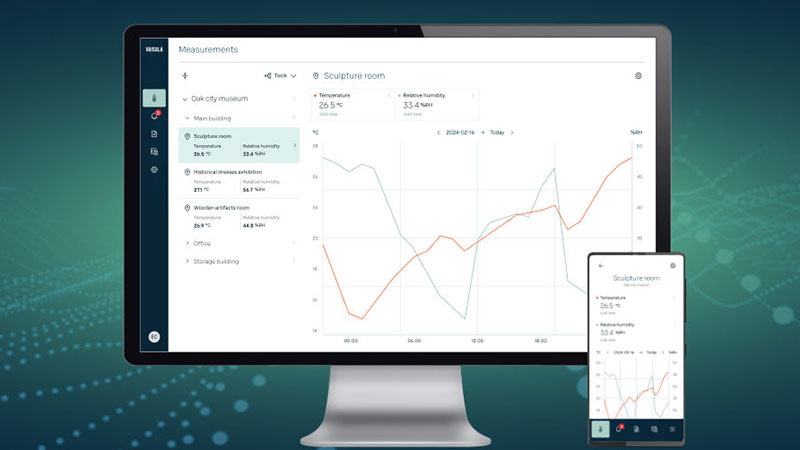
Jade Smart Cloud
Jade Smart Cloudは、産業向けの計測に安全で信頼性の高いワイヤレスモニタリングを組み合わせ、次世代の機能性と柔軟性をプロフェッショナルに提供します。

VDL200 データロガー
VDL200 データロガーは、ヴァイサラの優れたセンサ技術とモニタリングシステムソフトウェアを統合しています。

DL1016/1416 温度データロガー
ヴァイサラの多用途温度データロガーは、超低温フリーザー、冷凍・冷蔵庫、インキュベータの 4 用途の温度を 1 台でモニタリングします。

RFL100 VaiNet ワイヤレス温度データロガー
RFLシリーズのデータロガーでは、ヴァイサラのLoRa®ベースのVaiNet ワイヤレステクノロジー が使用されており、冷蔵庫、冷凍庫、インキュベータ、LN2タンク、コールドルーム、そして超低温冷凍庫の温度をモニタリングすることができます。各RFLデータロガーの屋内通信距離は100mです。

GMP252 CO₂プローブ
ヴァイサラCARBOCAP ® GMP252 CO 2 プローブは、ppmレベルのCO 2 計測用のインテリジェントプローブです。

HMT120/130 湿度温度変換器
ヴァイサラHUMICAP® HMT120 変換器およびHMT130 変換器は、クリーンルーム、博物館、研究所、データセンターでの使用に適しています。

HMD60シリーズ 湿度温度変換器
ヴァイサラ HUMICAP® HMD60シリーズ 湿度温度変換器は、堅牢で信頼性が高いダクト取り付けタイプの変換器で、要件の厳しい一般空調(HVAC)や軽工業用途での相対湿度のモニタリング向けに設計されています。

HMP1 湿度温度プローブ
ヴァイサラ HUMICAP® HMP1 湿度温度プローブは、屋内スペースの環境計測用に設計されています。

HMP9 極細湿度温度プローブ
ヴァイサラHUMICAP® HMP9 湿度温度プローブは、素早い応答性や計測性能、多くの化学物質への耐性が重要となる、急速に変化する環境に簡単に設置できるように設計されています。

