Why Vaisala is a preferred vendor for many semiconductor fabrication plants for qualifying H2O2 concentration in CMP of tungsten

Chemical mechanical planarization/polishing (CMP) is a critical, very costly, and challenging nano-polishing process that combines chemical reactions and mechanical abrasion. It is the key enabling step in integrated circuits manufacturing, affecting both yield and productivity.
CMP in a nutshell
Polishing is done with a slurry that includes an oxidizing agent, typically hydrogen peroxide (H2O2). During manufacturing, a wafer is pressed against a polishing pad while both wafer and pad are rotated counterclockwise at slightly different velocities. As slurry is dispensed on the center of the pad, the combination of mechanical and chemical actions gradually removes material from the wafer surface, leading to a surface that is locally and globally planar.
CMP slurries are mixed or diluted at the plant before use. Oxide polishing slurries are commonly purchased in concentrated form and diluted with water on-site to minimize shipping and labor costs. Some multicomponent polishing slurries may only be blended immediately prior to their use because of their short post-mix lifespan. The correct blend is essential as it is directly linked to chemical reaction rates and the wafer polishing rate; any defects in the blend have a negative impact on manufacturability and reliability. Although the slurry control at the point of manufacture (POM) is tight, subsequent processes – including transportation, handling, and filtration – can affect the chemical properties, and therefore continuous slurry monitoring is required until point of use (POU) to ensure high yield. Effective, rapid, reliable, precise, and cost-effective metrology tools and methodologies are required, so many fabrication plants choose to use refractometers.
How refractive index helps to improve production quality
Refractive index (RI) – a continuous, in-line, non-slurry-consuming measurement – helps fabrication plants identify slurry composition faults quickly as it conveys real-time information about the process, reducing the number of wafers at risk.
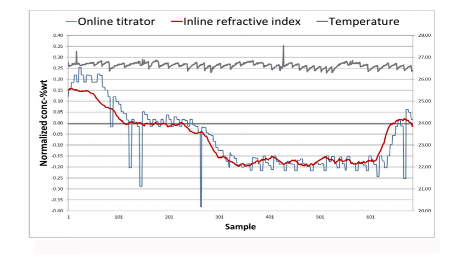
CMP slurry carries nanoparticles to solids content levels of 1–30% depending on the slurry type, making it challenging to analyze. However, once calibrated to a specific slurry’s temperature/RI characteristics, RI measurements can successfully determine the concentration of hydrogen peroxide in tungsten slurry to within ±0.03% by weight even in these difficult conditions.
Moreover, unlike conductivity probe tests, RI measurements can monitor H2O2 slurry density, an indicator of settling and degradation of the slurry over time. Therefore, the RI is used not only to qualify the final product but also to monitor batch-to-batch variations in the incoming raw slurry and to validate the blend-addition steps.
An attractive feature of some slurry delivery systems has been the ability to use an automated chemical-spiking function in the day tank.
Vaisala K-PATENTS® Semicon Refractometer benefits
The Vaisala K‑PATENTS Semicon Refractometer is specifically designed for semiconductor manufacturing environments. It has a small footprint and the construction is metal-free, making it ideal for measuring chemicals without corroding the process.
The Vaisala K‑PATENTS Semicon Refractometer is the superior choice for CMP operations because:
- Measurement is fully digital, and drift is impossible
- The integrated temperature measurement component ensures highly accurate RI measurement
- It offers direct density measurement
- The robust design tolerates process vibrations with no measurement errors
- Built-in diagnostics give an instant overview of process conditions
- Of the availability of different high-quality flow cells, that are designed to reduce and even eliminate fouling.
References
Diversified Fluid Solutions has been using the Vaisala K-PATENTS Semicon Refractometer successfully for years in its CMP operations and has found the device extremely reliable and accurate. “As advanced process nodes bring increasing numbers of CMP steps, we must ensure that the slurries delivered to the polishing tools maintain consistent chemical and mechanical characteristics,” explains Karl Urquhart, Director of R&D, Chemical Technology, Diversified Fluid Solutions. “Inline RI monitoring can evaluate the chemical composition of incoming material, qualify blend addition steps, and validate a uniform CMP slurry blend in a single real-time, non-slurry-consuming measurement.”
The first H2O2 measurement installation for CMP slurry was installed at a large semiconductor fabrication plant in 2013, replacing auto-titration. The measurement device has remained stable ever since, with no instrument maintenance required beyond routine flushing of the slurry blender.
In general, after installing a Vaisala K-PATENTS Semicon Refractometer the wafer throughput at fabrication plants increases by approximately 20%. In addition, CMP slurries are strictly controlled and enable improved uniformity of the planarization process.
Would you like to know more?
RI measurement is a simple, cost-effective, and accurate technique that provides real-time information on slurry composition, making a refractometer the metrology instrument of choice for many fabrication plants.
Contact us for more information.
Read more from our white paper: Inline Refractive Index Replaces Auto-titration in Qualifying H2O2 Concentration in CMP of Tungsten
All Vaisala’s applications in fab chemical process monitoring and fault detection







