Expert Article
Relative humidity (%RH) – How is it defined and calculated?
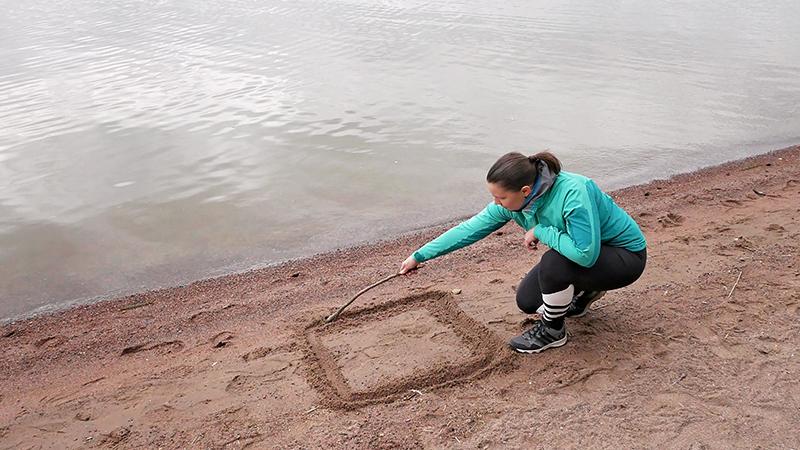

The first step on the journey to mastering humidity parameters is to understand partial pressures. We’ll begin our journey by the sea, with a one-meter square drawn in the sand. Now, imagine that there is an air column going up through the atmosphere and into space, with an air weight of about 10,300 kg. This air mass generates hydrostatic pressure with a force of 101,325 Newtons per square meter.
This defines a pressure unit called Pascal [Pa]: Newtons per square meter. That is the total pressure. The air we breathe is a mixture of gases, where the main components are nitrogen (N2), oxygen (O2), water vapor (H2O), argon (Ar), and carbon dioxide (CO2). These components can be expressed using Dalton’s law of partial pressures:
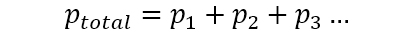
The component with the concentration that varies the most is water vapor, and it plays an important role in affecting weather and climate, as well as numerous industrial processes and other aspects of our everyday lives.
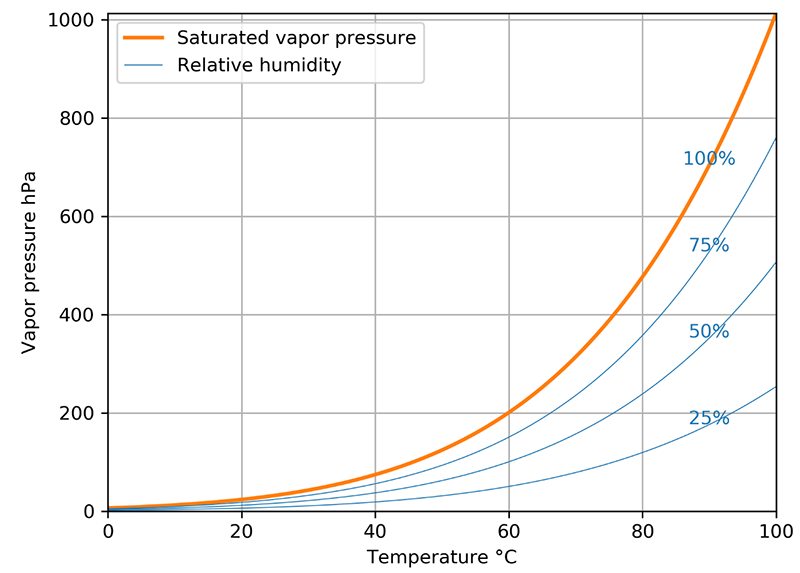
On earth, water exists in three different forms: ice, water, and gas. The maximum amount of gaseous water is dictated by the temperature: the higher the temperature, the higher the partial pressure of water vapor, i.e. there is more dissolved water in the air. The maximum water vapor pressure at a specific temperature is called water vapor saturation pressure. Water vapor pressure cannot exceed this limit, and a simple way to prove this fact is to look at the clouds in the sky. Clouds are formed when the water vapor saturation pressure has been reached and the environment can no longer hold the water in its vaporous phase, meaning some of it condenses into tiny droplets that may eventually fall on us as rain.
Relative humidity (%RH) is a concept that defines what percentage of water vapor is present at that temperature relative to the saturation point. When it’s raining, it’s obviously 100 %RH up there in the clouds where the droplets are forming. While the instruments and technologies available to us have greatly improved over the years, the weather can still sometimes surprise us. We humans tend to try to control our environment, and this has led to great strides forward in technological and industrial development. We can create controlled environments depending on our need, whether it’s for laundry drying or manufacturing cutting-edge technology from highly sensitive materials.
What do a wet cotton shirt, conducting salts used for lithium battery manufacturing, and a HUMICAP® thin-film polymer sensor have in common? The answer is they are all hygroscopic materials, which means that they attract water molecules from their surrounding environment until they reach an equilibrium state. Here, relative humidity (RH) plays an important role because the equilibrium moisture content of a material is closely related to the relative humidity level. Luckily, I can trust that the battery in my mobile phone has been manufactured in a controlled environment and protected in a way that it tolerates occasional splashes. What about the cotton shirt that got wet in the rain? It doesn’t dry that well outdoors while it’s raining – even if I protect it from the rain. However, I can greatly enhance the shirt’s drying rate by putting it into a clothes dryer. Why does the shirt dry faster at a high temperature? Is there less humidity in the drying cabinet? The short answer is that the air is relatively drier at elevated temperatures. This means that the relative humidity (RH) inside the dryer is lower and the cotton shirt therefore seeks to achieve equilibrium with its surroundings and eventually becomes drier.
Relative humidity (RH) is the ratio of the water vapor pressure to the saturation vapor pressure at a given temperature. If you want to deepen your knowledge of how to calculate relative humidity, related quantities, and many other humidity parameters, download our Conversion Formulas technical eBook.

Joni Partanen is a Product Manager at Vaisala. He is responsible for the development of humidity and vaporized hydrogen peroxide measuring products. He has over 16 years of experience in process industry measurement technology, industrial engineering, and instrumentation. Joni holds a Bachelor of Engineering degree in Automation Technology.
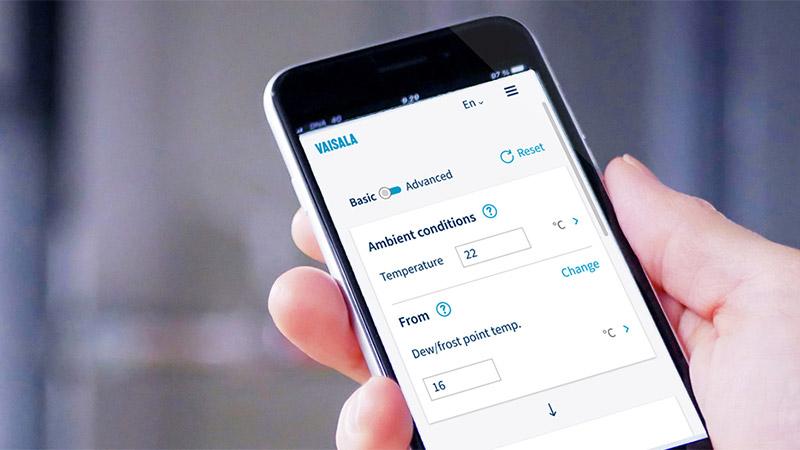
Vaisala humidity calculator
Relative humidity, absolute humidity, wet-bulb temperature, enthalpy, water concentration, and many more. Humidity calculations and conversions made easy.
Relative humidity related products

Humidity and Temperature Transmitter Series HMDW80

Moisture and Temperature Meter Series MMT310

Hand-Held Humidity and Temperature Meter HM40

Humidity and Temperature Transmitters HMT120/130

Turbine Mounting Kit HMT300TMK

Humidity and Temperature Probe HMP110

HUMICAP® Humidity and Temperature Probe HMP113

Digital Open Frame Humidity Module HMM105

Humidity Module HMM100

HMP60 Humidity and Temperature Probe

INTERCAP® Humidity and Temperature Probe HMP63

Relative Humidity and Temperature Probe HMP7

Relative Humidity and Temperature Probe HMP4

Relative Humidity and Temperature Probe HMP5

Relative Humidity and Temperature Probe HMP8
