Expert article
Understanding humidity, part 1: Fundamental concepts every engineer should know


Humidity sounds simple in theory – after all, it’s just a measure of water vapor in air. But not everyone understands the relationships between different humidity parameters, or how humidity changes with temperature and pressure. This article aims to demystify key humidity parameters and explain why they matter in different industrial applications.
Why is understanding humidity important?
Most engineers can measure humidity, but not everyone understands how different humidity parameters relate to one another, or how they change with temperature and pressure. Getting this wrong – sometimes even if it’s a seemingly small mistake – can lead to significant process impacts, such as poor product quality, wasted energy, or non-compliance.
The consequences of inaccurate humidity measurements can vary depending on the application. Here are some examples of applications and the potential problems that inaccurate measurement can cause:
- HVAC & building automation: reduced comfort, lower indoor air quality, poor energy efficiency
- Cleanrooms (pharma, biotech, semiconductor): regulatory non-compliance, risks to product safety
- Semiconductor manufacturing: lowered manufacturing yield
- Battery production and dry rooms: safety risks, poor performance, reduced manufacturing yield
- Food and beverage: poor product consistency, contamination
- Compressed air systems: condensation and corrosion
Key humidity concepts every engineer should know
From overdrying and increased energy costs to underestimating condensation risk and product spoilage, no matter the industry, misinterpreting humidity levels will lead to poor control decisions. How can you measure humidity accurately? Here’s a simple explainer of what you need to know.
Relative humidity (RH)
RH is the most commonly used humidity unit, but it is still commonly misunderstood. RH is strongly dependent on temperature – the ‘relative’ refers to the difference between the amount of water vapor present and the maximum amount that is physically possible for the air to hold at that temperature. RH is expressed as a percentage: the partial water vapor pressure in relation to the saturation pressure.
pw = partial water vapor pressure
pws = water vapor’s saturation pressure
If RH reaches 100%, the maximum amount of water the air can hold has been reached – if more is introduced, an equal amount of water must transform back into liquid water or ice through condensation. When there is no water vapor in the air, the RH is 0% regardless of the temperature. Because the saturation pressure depends strongly on temperature, as the temperature rises, saturation pressure goes up. This means that even if the moisture content stays the same, RH will drop as the temperature rises.
- RH in the real world: The outside temperature is -14 °C and the relative humidity is 60%. As the air enters an office building it is heated to +21 °C, but the amount of water remains constant – no water is added or removed from the air in normal ventilation systems. Because of the heating, the saturation pressure of the water vapor rises, meaning the maximum possible amount of water vapor in the air increases. As the partial pressure of the water vapor is unchanged, the RH will drop to 5%, which is usually considered too dry for comfort.
- Why relying on RH alone can be a mistake: RH is highly dependent on temperature, so even small temperature changes can cause large swings in RH without any actual change in moisture content. This is because RH tells you how close the air is to saturation at the current temperature – not how much water is actually present. Because of this, RH can be misleading if used as a standalone parameter. In very dry, pressurized environments such as compressed air systems, RH is practically useless because all the relevant values are extremely low (often below 1% RH), meaning they offer poor resolution and no meaningful differentiation in compressed air quality.
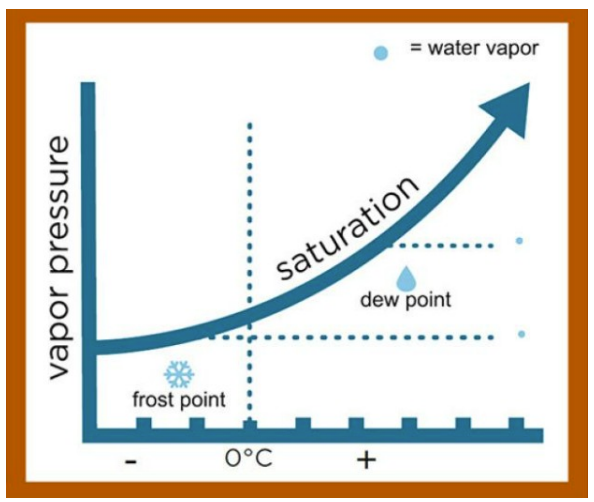
Dew point (Td) and frost point (Tf)
Dew point temperature is the second most commonly used humidity parameter. Simply put, it is the temperature to which air must be cooled to become saturated with water vapor. At this point, any additional water will begin condensing. Unlike RH, dew point temperature is independent of ambient temperature. It correlates with the amount of water in the air and is always less than or equal to the actual temperature.
When the dew point temperature is below 0 °C, it is more precisely called frost point (Tf), because moisture deposits as ice rather than liquid water. In practice, the terms are often used interchangeably, and instruments typically report a combined "dew/frost point" (Td/f) value.
Dew point temperature is affected by pressure, with higher pressure raising the dew point temperature. Under normal atmospheric conditions, the dew point temperature cannot exceed 100 °C, because at this temperature the air would consist entirely of water vapor. To increase the amount of water beyond this value, vapor density and therefore pressure must rise. In specialized applications such as semiconductor processes, where vacuum is used to improve material drying, the dew point can be as low as –80 °C, which corresponds to about 1 ppm of water vapor.
The water vapor saturation pressure at different temperatures is a known variable, so the dew point can be calculated from RH and temperature. Conversely, if dew point and either temperature or RH are known, the missing variable can be calculated. Dew point is the most reliable measurement at low humidity levels. Measurement uncertainties propagate into calculated humidity parameters. At very low humidity levels, it is therefore often more accurate to measure the dew point directly, since dew point calculated from RH and temperature can be significantly less precise.
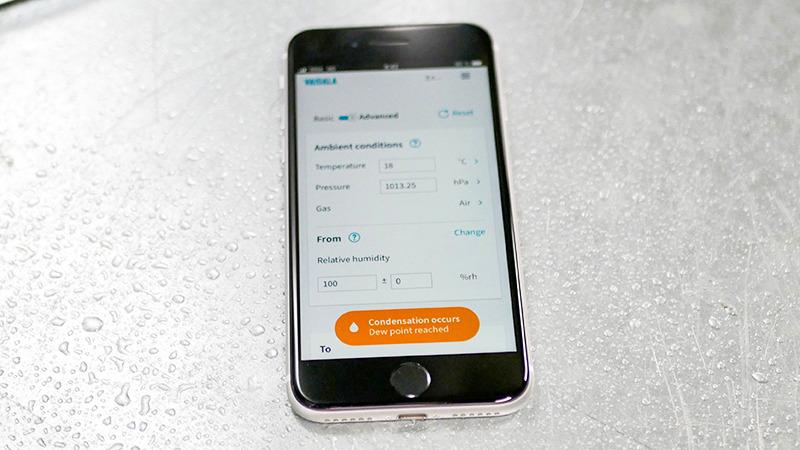
Dew point in the real world: In a cleanroom, the control target is 40 (±2) % RH at a temperature of 20 (±1) °C . Because RH depends on temperature it is not the best control parameter here – it would be virtually impossible to dry or humidify the space while trying to maintain a stable temperature. The solution is to use dewpoint temperature as the control parameter instead. At 40 % RH and 20 °C the dewpoint is 6.0 °C. A narrow dewpoint control band makes environmental control easier and saves energy.
- Why dew/frost point beats RH in demanding applications: In very dry, pressurized environments such as compressed air systems, RH is practically useless — all values fall below 1 % RH, offering poor resolution and no meaningful differentiation. Td/f provides a standardized, actionable measure of moisture content and directly indicates the temperature at which condensation (or ice formation) will occur under system pressure. This is critical for preventing problems such as freezing in pressure lines, water hammering, gasket failures, and lubricant washout. Td/f is also the metric referenced in compressed air standards, ensuring compliance.
Absolute humidity (a)
Absolute humidity describes how many grams of water vapor are present per cubic meter of air. Because it provides a reliable measurement of the amount of water present, absolute humidity is a fairly widely used parameter, especially in drying or process control applications where the actual water mass is more important that the percentage saturation.
The density of air varies with pressure, so the absolute humidity depends quite strongly on the gas pressure. In pressurized processes, the pressure must be known in order to calculate absolute humidity from the other humidity variables.
Enthalpy (h)
Enthalpy is the total energy content of moist air compared to a reference state. It represents the amount of energy needed to heat dry air to its current temperature from
0 °C. While enthalpy isn’t strictly a humidity measurement, water vapor has a very high specific heat capacity and can be present in air in very different concentrations, meaning it has a strong influence on enthalpy.
Enthalpy is most commonly used when comparing the heat content of gases in heating, ventilation, and air conditioning (HVAC) systems. It is important to remember that when enthalpy is expressed in imperial units the reference point will be different. Therefore, enthalpy values calculated in different units are not comparable.
Mixing ratio (x)
The mixing ratio defines the mass of water vapor in the volume occupied by one kilogram of dry gas. The density of air varies with pressure, so the mixing ratio is also dependent on the pressure of the gas. In pressurized processes, the pressure must be known in order to calculate the mixing ratio from other humidity variables.
The mixing ratio is mainly used for calculating water content when the mass flow of air is known, for example in ventilation systems.
Pressure effect
Dalton’s law states that a change in the total pressure of a gas must have an effect on the partial pressures of all of the component gases, including water vapor. If, for example, the total pressure is doubled, the partial pressures of all component gases are doubled as well.
In compressed air applications, increasing the pressure removes water from the air. This happens because the partial pressure of water vapor (pw) is increased, but the saturation pressure is still only a function of temperature. As the pressure builds in a receiver tank and pw reaches pws, water condenses into liquid and ultimately must be drained from the tank. Ignoring the pressure in pressurized systems can lead to underestimating the condensation risk.
When you understand how different humidity parameters relate to one another and how they change with temperature and pressure, you can avoid small mistakes that can have significant process impacts like poor product quality, wasted energy, or non-compliance. In part 2 of this series you’ll learn how humidity behaves in real-world conditions and how to choose the right humidity measurement instrument for your application. Check also our website for more information about humidity.
Want to go deeper into humidity measurement theory? Download our comprehensive Humidity Measurement eGuide for smart industries.

Vuokko Lantz represents Global Product Management at Vaisala Industrial Measurements. She has more than two decades of experience on the development and application of sensor technologies in various industrial and consumer applications. Vuokko holds Doctor of Science (Computer and Information Science, Usability Research) and Master of Science (System Analysis and Operation Research) degrees in Technology.
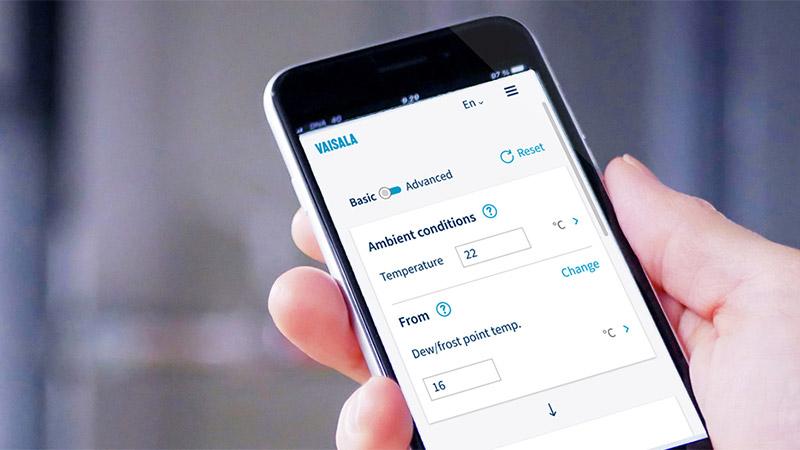
Vaisala humidity calculator
Relative humidity, absolute humidity, wet-bulb temperature, enthalpy, water concentration, and many more. Humidity calculations and conversions made easy.